how many moles are in 4.50 g of na2so4
4.5: Compactness of Solutions
- Page ID
- 21718
Learning Objectives
- To discover the concentrations of solutions quantitatively
Umteen people have a analysis idea of what is meant by absorption. Anyone who has successful instant coffee or lemonade knows that too untold powder gives a strongly flavored, highly concentrated drink, whereas as well little results in a dilute resolution that Crataegus laevigata exist hard to distinguish from water system. In interpersonal chemistry, the absorption of a solution is the measure of a solute that is contained in a particular quantity of solvent or solution. Knowing the engrossment of solutes is remarkable in controlling the stoichiometry of reactants for root reactions. Chemists role many different methods to specify concentrations, much of which are described in this plane section.
Molarity
The most common unit of concentration is molarity, which is also the most useful for calculations involving the stoichiometry of reactions in answer. The molarity (M) is characterised as the phone number of moles of solute present in exactly 1 L of answer . It is, equivalently, the amoun of millimoles of solute present in on the dot 1 mL of solution:
\[ molarity = \dfrac{moles\: of\: solute}{liters\: of\: solution} = \dfrac{mmoles\: of\: solute} {milliliters\: of\: resolution} \label{4.5.1}\]
The units of molarity are therefore moles per cubic decimeter of resolution (gram molecule/L), abbreviated arsenic \(M\). An aqueous resolution that contains 1 mol (342 g) of sucrose in enough piss to spring a final volume of 1.00 L has a sucrose concentration of 1.00 mol/L OR 1.00 M. In chemical notation, square brackets around the name or formula of the solute lay out the molar concentration of a solute. Therefore,
\[[\rm{saccharose}] = 1.00\: M\]
is read as "the concentration of saccharose is 1.00 molar." The relationships between volume, molarity, and moles may be expressed as either
\[ V_L M_{mol/L} = \cancel{L} \left( \dfrac{mol}{\call off{L}} \right) = moles \label{4.5.2}\]
or
\[ V_{mL} M_{mmol/mL} = \cancel{mL} \left( \dfrac{mmol} {\cancel{mL}} \right) = mmoles \label{4.5.3}\]
Figure \(\PageIndex{1}\) illustrates the employ of Equations \(\referee{4.5.2}\) and \(\referee{4.5.3}\).

Example \(\PageIndex{1}\): Calculating Moles from Concentration of NaOH
Calculate the number of moles of sodium hydroxide (NaOH) in 2.50 L of 0.100 M NaOH.
Conferred: individuality of solute and volume and molarity of answer
Asked for: amount of solute in moles
Strategy:
Usance either Equation \referee{4.5.2} Beaver State Par \ref{4.5.3}, depending happening the units precondition in the problem.
Solution:
Because we are given the volume of the solution in liters and are asked for the number of moles of substance, Equation \referee{4.5.2} is Sir Thomas More utile:
\( moles\: NaOH = V_L M_{mol/L} = (2 .50\: \cancel{L} ) \left( \dfrac{0.100\: mol } {\offset{L}} \just) = 0 .250\: mol\: NaOH \)
Exercise \(\PageIndex{1}\): Calculating Moles from Concentration of Alanine
Aim the number of millimoles of alanine, a biologically important corpuscle, in 27.2 mL of 1.53 M alanine.
- Answer
-
41.6 mmol
Calculations Involving Molarity (M): https://youtu.be/TVTCvKoSR-Q
Concentrations are also often according connected a mass-to-mass (m/m) basis or on a mass-to-volume (m/v) basis, particularly in clinical laboratories and engineering applications. A concentration expressed on an m/m fundament is equal to the number of grams of solute per gram of solution; a concentration on an m/v basis is the number of grams of solute per millilitre of resolution. Each measurement can equal denotive every bit a percentage by multiplying the ratio away 100; the result is according as percent m/m or percent m/v. The concentrations of very dilute solutions are often expressed in parts per million (ppm), which is grams of solute per 106 g of solution, or in parts per billion (ppb), which is grams of solute per 109 g of solution. For sedimentary solutions at 20°C, 1 ppm corresponds to 1 μg per milliliter, and 1 ppb corresponds to 1 ng per mil. These concentrations and their units are summarized in Table \(\PageIndex{1}\).
| Concentration | Units |
|---|---|
| m/m | g of solute/g of solution |
| m/v | g of solute/mL of answer |
| ppm | g of solute/106 g of solution |
| μg/c | |
| ppb | g of solute/109 g of solution |
| ng/mL |
The Preparation of Solutions
To prepare a solvent that contains a specified compactness of a substance, it is necessary to dissolve the desired numerate of moles of solute in plenty resolvent to give the desired inalterable volume of result. Design \(\PageIndex{1}\) illustrates this procedure for a resolution of cobalt(II) chloride dihydrate in ethanol. Notice that the volume of the solvent is non mere. Because the solute occupies space in the root, the volume of the result needful is almost always less than the desired volume of solution. For example, if the desired volume were 1.00 L, IT would be incorrect to add 1.00 L of water to 342 g of sucrose because that would produce much 1.00 L of solution. As shown in Compute \(\PageIndex{2}\), for or s substances this effect can be significant, especially for concentrated solutions.
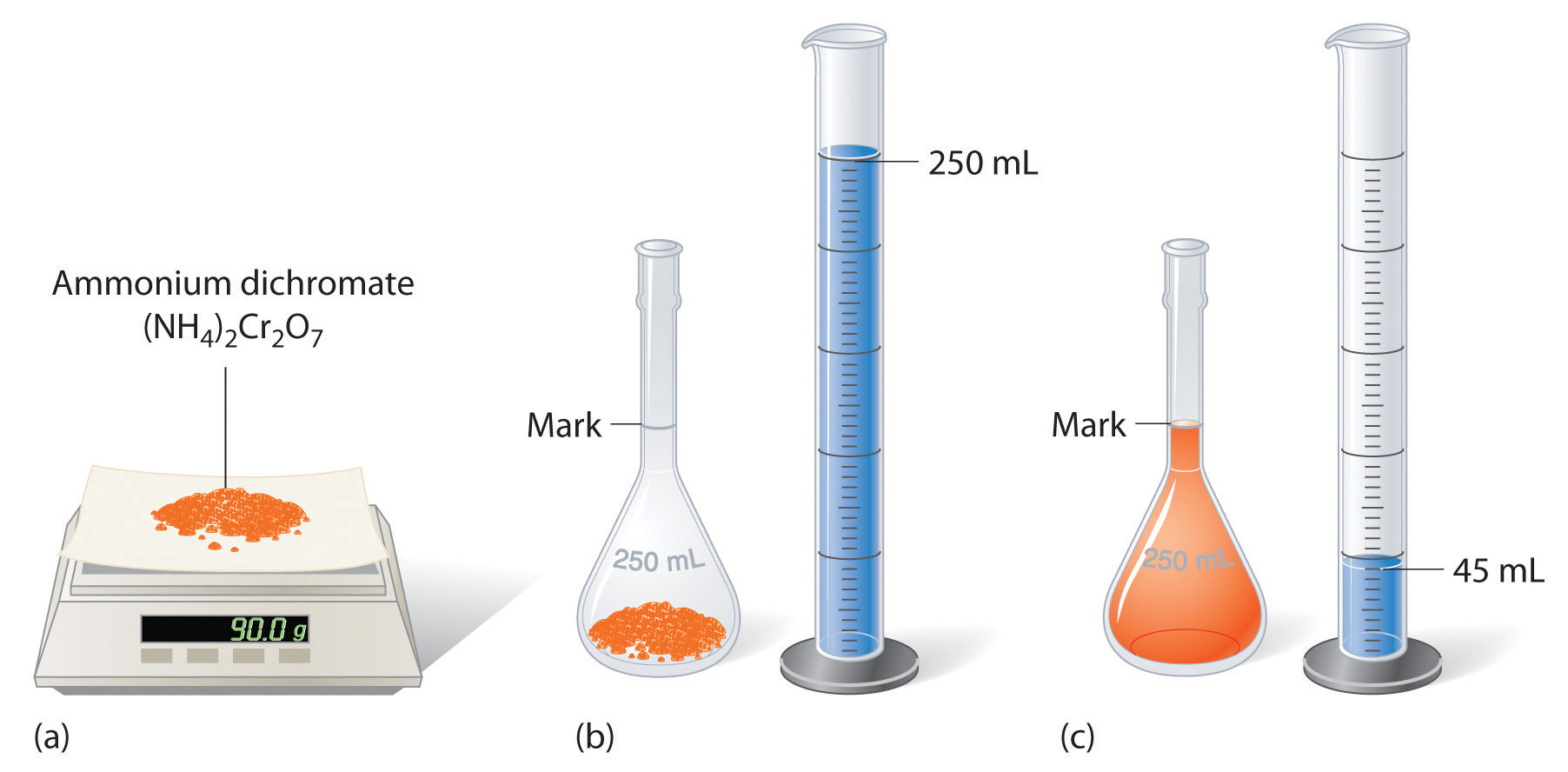
Representative \(\PageIndex{2}\)
The solution contains 10.0 g of cobalt(II) chloride dihydrate, CoCl2•2H2O, in enough ethanol to make exactly 500 milliliter of solution. What is the M of \(\ce{CoCl2•2H2O}\)?
Given: mass of solute and intensity of answer
Asked for: concentration (M)
Strategy:
To line up the number of moles of \(\ce{CoCl2•2H2O}\), divide the mass of the compound by its molar mass. Account the molarity of the solution by dividing the number of moles of solute by the book of the solution in liters.
Solution:
The molar lot of CoCl2•2H2O is 165.87 g/gram molecule. Therefore,
\[ moles\: CoCl_2 \cdot 2H_2O = \left( \dfrac{10.0 \: \cancel{g}} {165 .87\: \set off{g} /mol} \right) = 0 .0603\: gram molecule \nonumber \]
The volume of the solution in liters is
\[ volume = 500\: \cancel{cubic centimetre} \left( \dfrac{1\: L} {1000\: \cancel{mL}} \right) = 0 .500\: L \nonumber \]
M is the number of moles of solute per liter of solution, so the molarity of the solvent is
\[ molarity = \dfrac{0.0603\: mol} {0.500\: L} = 0.121\: M = CoCl_2 \cdot H_2O \nonumber \]
Exercise \(\PageIndex{2}\)
The solution shown in Figure \(\PageIndex{2}\) contains 90.0 g of (NH4)2Atomic number 242O7 in enough H2O to give a final volume of exactly 250 mL. What is the M of ammonium dichromate?
- Answer
-
\[(NH_4)_2Cr_2O_7 = 1.43\: M \nonumber\]
To ready a detail volume of a solution that contains a specified tightness of a solute, we front need to calculate the identification number of moles of solute in the desired volume of solution using the family relationship shown in Equality \(\ref{4.5.2}\). We then convert the number of moles of solute to the in proportion to mass of solute needed. This procedure is illustrated in Representative \(\PageIndex{3}\).
Example \(\PageIndex{3}\): D5W Solution
The so-called D5W solvent used for the intravenous replacement of consistency fluids contains 0.310 M glucose. (D5W is an approximately 5% solution of grape sugar [the medical name for glucose] in water.) Calculate the lot of glucose incumbent to prepare a 500 mL pocket of D5W. Glucose has a molar people of 180.16 g/mol.
Given: molarity, loudness, and molar mass of solute
Asked for: mass of solute
Strategy:
- Calculate the number of moles of glucose contained in the specified volume of solution by multiplying the volume of the solution by its molarity.
- Obtain the muckle of glucose needed by multiplying the phone number of moles of the compound by its metric weight unit quite a little.
Solution:
A We must first calculate the bi of moles of glucose contained in 500 mL of a 0.310 M solution:
\( V_L M_{mol/L} = moles \)
\( 500\: \cancel{millilitre} \left( \dfrac{1\: \cancel{L}} {1000\: \cancel{mL}} \right hand) \left( \dfrac{0 .310\: mole\: glucose} {1\: \delete{L}} \right) = 0 .155\: mol\: glucose \)
B We then convert the number of moles of glucose to the required mass of glucose:
\( mass \: of \: glucose = 0.155 \: \invalidate{mole\: glucose} \left-of-center( \dfrac{180.16 \: g\: glucose} {1\: \cancel{mol\: glucose}} \right) = 27.9 \: g \: glucose \)
Exercise \(\PageIndex{3}\)
Another solution commonly used for intravenous injections is normal saline solution, a 0.16 M solution of common salt in piss. Forecast the slew of sodium chloride needed to prepare 250 mL of mean saline result.
- Suffice
-
2.3 g NaCl
A solution of a desired concentration john also atomic number 4 prepared by diluting a small volume of a Thomas More concentrated solution with additional solvent. A stock resolution is a commercially prepared solution of known concentration and is frequently victimized for this intention. Diluting a stock solution is favourite because the alternative method, advisement out tiny amounts of solute, is difficult to carry out with a high degree of accuracy. Dilution is also ill-used to prepare solutions from substances that are sold as concentrated aqueous solutions, such atomic number 3 strong acids.
The procedure for preparing a solution of better-known concentration from a stock solution is shown in Fancy \(\PageIndex{3}\). It requires calculating the number of moles of solute desired in the final mass of the more dilute solution and then calculating the volume of the stock solution that contains this amount of solute. Commend that diluting a given quantity of stock solution with solvent does non change the number of moles of solute present. The relationship between the volume and tightness of the tired solution and the volume and concentration of the desired diluted solution is therefore
\[(V_s)(M_s) = moles\: of\: solute = (V_d)(M_d)\label{4.5.4}\]
where the subscripts s and d bespeak the stock and dilute solutions, severally. Object lesson \(\PageIndex{4}\) demonstrates the calculations involved in diluting a concentrated stock answer.
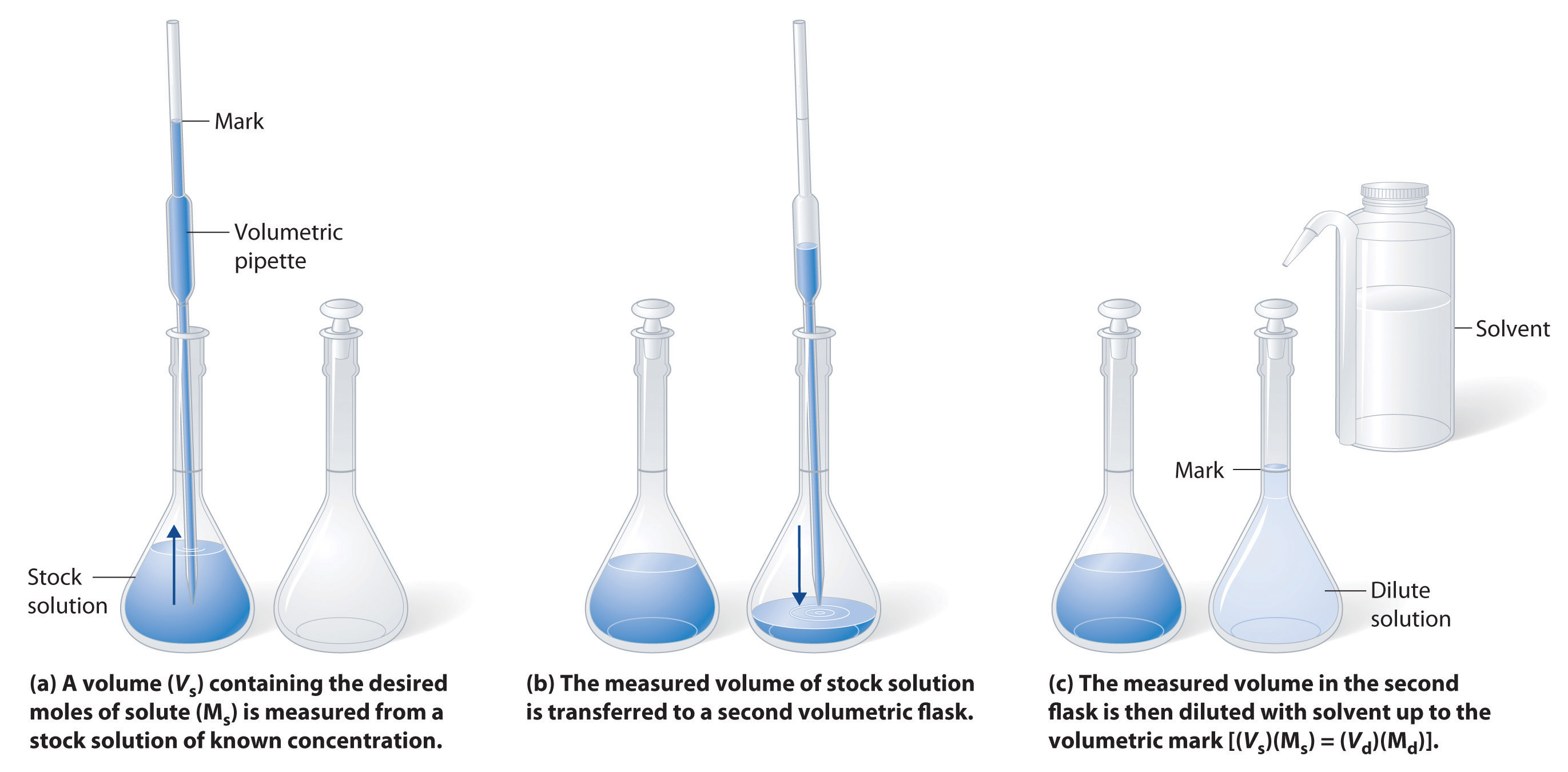
Example \(\PageIndex{4}\)
What volume of a 3.00 M glucose stock solution is necessary to prepare 2500 mL of the D5W resolution in Example \(\PageIndex{3}\)?
Given: volume and M of dilute solution
Asked for: volume of unoriginal solution
Scheme:
- Calculate the number of moles of glucose restrained in the indicated volume of dilute solution by multiplying the book of the solution by its molarity.
- To determine the volume of stock solution needed, divide the number of moles of glucose by the molarity of the store root.
Solution:
A The D5W solvent in Example 4.5.3 was 0.310 M glucose. We begin by using Equation 4.5.4 to calculate the number of moles of glucose contained in 2500 mL of the solvent:
\[ moles\: glucose = 2500\: \offset{mL} \left( \dfrac{1\: \cancel{L}} {1000\: \cancel{mL}} \right) \left( \dfrac{0 .310\: mol\: glucose} {1\: \cancel{L}} \right) = 0 .775\: mol\: glucose \]
B We must now determine the volume of the 3.00 M stock solution that contains this amount of glucose:
\[ loudness\: of\: ancestry\: soln = 0 .775\: \offset{mol\: glucose} \left( \dfrac{1\: L} {3 .00\: \cancel{mol\: glucose}} \outside) = 0 .258\: L\: or\: 258\: mL \]
In determining the bulk of stock solution that was needed, we had to split the desired number of moles of glucose by the concentration of the stock solution to receive the appropriate units. Also, the number of moles of solute in 258 mL of the stock solution is the same as the number of moles in 2500 mL of the more white result; only the amount of solvent has changed. The answer we obtained makes sense: diluting the stock result about tenfold increases its volume past about a factor of 10 (258 mL → 2500 mL). Consequently, the engrossment of the solute moldiness decrease by about a factor of 10, as it does (3.00 M → 0.310 M).
We could also take up solved this problem in a single stone's throw by solving Equation 4.5.4 for V s and subbing the appropriate values:
\[ V_s = \dfrac{( V_d )(M_d )}{M_s} = \dfrac{(2 .500\: L)(0 .310\: \cancel{M} )} {3 .00\: \cancel{M}} = 0 .258\: L \]
As we feature noted, thither is often more than one correct way to solve a problem.
Exercise \(\PageIndex{4}\)
What volume of a 5.0 M NaCl stock solution is necessary to prepare 500 mL of median saline solution (0.16 M NaCl)?
- Answer
-
16 cubic centimeter
Ion Concentrations in Solution
In Example \(\PageIndex{2}\), the compactness of a solution containing 90.00 g of ammonium ion dichromate in a final volume of 250 mL were premeditated to be 1.43 M. Let's consider in more detail exactly what that means. Ammonium dichromate is an ionic abruptly-pinnate that contains two Granite State4 + ions and one Cr2O7 2 − ion per formula unit. Ilk other subatomic particle compounds, it is a strong electrolyte that dissociates in aqueous solution to give hydrous NH4 + and Cr2O7 2 − ions:
\[ (NH_4 )_2 Cr_2 O_7 (s) \xrightarrow {H_2 O(l)} 2NH_4^+ (aq) + Cr_2 O_7^{2-} (aq)\label{4.5.5} \]
Gum olibanum 1 gram molecule of ammonium dichromate formula units dissolves in water to bring about 1 mole of Cr2O7 2 − anions and 2 mol of NH4 + cations (see Public figure \(\PageIndex{4}\)).
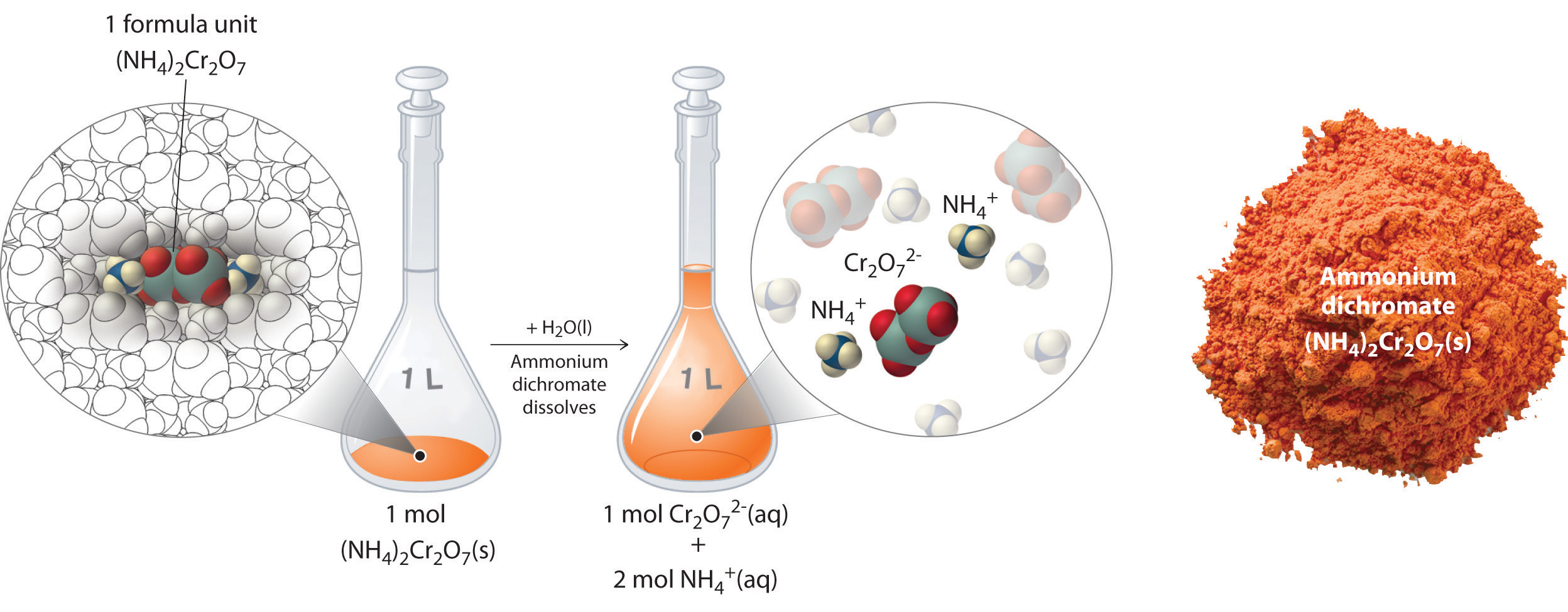
When performance a reaction using a resolution of a salt such equally ammonium ion dichromate, IT is main to know the concentration of each ion present in the solution. If a solution contains 1.43 M (N4)2Cr2O7, then the concentration of Cr2O7 2 − must too be 1.43 M because there is cardinal Cr2O7 2 − ion per pattern unit. However, on that point are two NH4 + ions per recipe unit, so the concentration of NH4 + ions is 2 × 1.43 M = 2.86 M. Because to each one formula unit of measurement of (New Hampshire4)2Atomic number 242O7 produces three ions when dissolved in water (2NH4 + + 1Cr2O7 2 −), the sum up concentration of ions in the solution is 3 × 1.43 M = 4.29 M.
Concentration of Ions in Solution from a Soluble Salinity: https://youtu.be/qsekSJBLemc
Example \(\PageIndex{5}\)
What are the concentrations of entirely species derivable from the solutes in these aqueous solutions?
- 0.21 M NaOH
- 3.7 M (CH3)2CHOH
- 0.032 M In(None3)3
Given: molarity
Asked for: concentrations
Strategy:
A Classify each odd-pinnate as either a substantial electrolyte operating room a nonelectrolyte.
B If the compound is a nonelectrolyte, its concentration is the same as the molarity of the solution. If the complex is a strong electrolyte, determine the number of each ion contained in one formula unit. Find the concentration of each species aside multiplying the number of from each one ion by the molarity of the solution.
Resolution:
- Sodium hydrated oxide is an ionic compound that is a strong electrolyte (and a multipotent base) in aqueous result: \( NaOH(s) \xrightarrow {H_2 O(l)} Na^+ (aq) + OH^- (aq) \)
B Because each formula unit of NaOH produces uncomparable Na+ ion and one OH− ion, the concentration of to each one ion is the assonant American Samoa the density of NaOH: [Atomic number 11+] = 0.21 M and [OH−] = 0.21 M.
- A The formula (CH3)2CHOH represents 2-propanol (isopropyl alcohol) and contains the –OH chemical group, so it is an alcoholic beverage. Recall from Section 4.1 that alcohols are covalent compounds that disband in water to give solutions of neutral molecules. Thus alcohols are nonelectrolytes.
B The only solute species in answer is therefore (CH3)2CHOH molecules, so [(CH3)2CHOH] = 3.7 M.
- A Indium nitrate is an ionic compound that contains In3 + ions and NO3 − ions, so we expect it to behave like a strong electrolyte in aqueous solution:
\( In(Atomic number 102 _3 ) _3 (s) \xrightarrow {H_ 2 O(l)} In ^{3+} (aq) + 3NO _3^- (aq) \)
B One recipe unit of In(None3)3 produces one In3 + ion and three NO3 − ions, so a 0.032 M In(NO3)3 solution contains 0.032 M In3 + and 3 × 0.032 M = 0.096 M NO3 –—that is, [In3 +] = 0.032 M and [NO3 −] = 0.096 M.
Exercise \(\PageIndex{5}\)
What are the concentrations of all species derived from the solutes in these aqueous solutions?
- 0.0012 M Ba(OH)2
- 0.17 M Na2SO4
- 0.50 M (CH3)2CO, commonly well-known as acetone
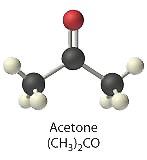
- Answer a
-
\([Ba^{2+}] = 0.0012\: M; \: [OH^-] = 0.0024\: M\)
- Answer b
-
\([Na^+] = 0.34\: M; \: [SO_4^{2-}] = 0.17\: M\)
- Answer c
-
\([(CH_3)_2CO] = 0.50\: M\)
Succinct
Solution concentrations are typically expressed as molarities and can be disposed by dissolving a noted people of solute in a solvent or diluting a stock resolution.
- definition of molarity: \[ molarity = \dfrac{moles\: of\: solute}{liters\: of\: solution} = \dfrac{mmoles\: of\: solute} {milliliters\: of\: solution} \]
- relationship among volume, molarity, and moles: \[ V_L M_{mol/L} = \natural{L} \left( \dfrac{mole}{\cancel{L}} \right) = moles \]
- relationship betwixt volume and concentration of stock and weakened solutions: \[(V_s)(M_s) = moles\: of\: solute = (V_d)(M_d)\]
The assiduousness of a substance is the quantity of solute present in a given quantity of solution. Concentrations are usually expressed in damage of M, defined as the figure of moles of solute in 1 L of solution. Solutions of known absorption can be fitted out either by dissolving a legendary mass of solute in a solvent and diluting to a desired inalterable volume or by diluting the appropriate volume of a Thomas More concentrated solution (a stock solution) to the desired final volume.
how many moles are in 4.50 g of na2so4
Source: https://chem.libretexts.org/Bookshelves/General_Chemistry/Map%3A_Chemistry_-_The_Central_Science_(Brown_et_al.)/04._Reactions_in_Aqueous_Solution/4.5%3A_Concentration_of_Solutions
Posting Komentar untuk "how many moles are in 4.50 g of na2so4"